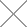Pressure Definition, Units & Conversion
What is Pressure?
Pressure generally refers to the amount of force that is exerted on a certain area. The formula of pressure is usually represented by p= F/A. ‘F’ refers to the applied force, and ‘A’ is the area of the region.
Units of pressure
Here are the most commonly used units of pressure in various systems:

1. SI Unit of Pressure(International System of Units)
Pascal (Pa): This is the standard unit of pressure in the SI system. And we also use mPa (millipascal). It is a subunit of the Pascal. And kPa. They are commonly used when measuring very small pressures, such as in certain scientific and engineering applications.
1 MPa = 1,000 kPa = 1,000,000 Pa
2. CGS Unit (Centimeter-Gram-Second System)
Barye (Ba): Barye is not commonly used. It is a unit of pressure in the centimeter-gram-second (CGS) system of units, which is less commonly used today compared to the International System of Units (SI).
1 barye is equal to 1 dyne per square centimeter (dyn/cm²).
3. Imperial Units
Pounds per Square Inch (psi). Commonly used in the United States, 1 psi is the pressure resulting from a force of one pound-force applied to an area of one square inch.
4. Other Units
Atmosphere (atm): This is a unit of pressure commonly used in various contexts. Standard atmospheric pressure at sea level is approximately 101,325 Pa or 1 atm.
Torr: Named after Evangelista Torricelli, 1 torr is equal to 1 mmHg (millimeter of mercury).
Bar: 1 bar is equal to 100,000 Pa.
Technical Atmosphere (at): commonly used in certain fields, where 1 at is defined as 1 kgf/cm².
FAQs
1. How to convert between these pressure units?
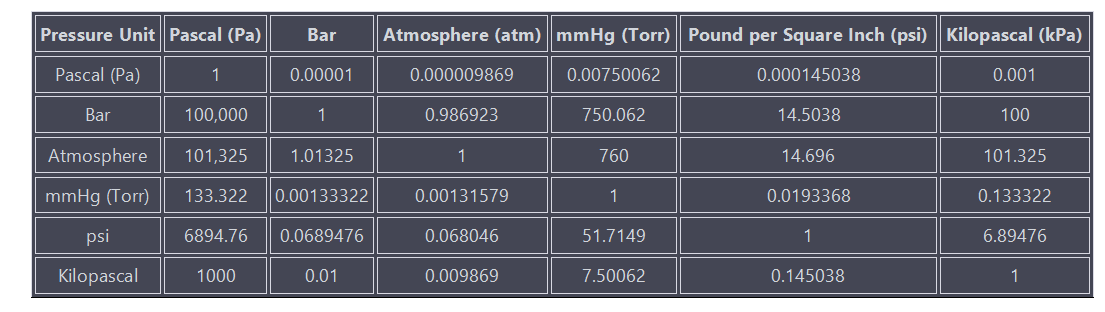
Please note that the values in the table are rounded and approximate. And you can also use the conversion tool.
2. How do we measure pressure?
Pressure values can be stated in three ways: Relative, absolute and differential. To measure these pressure values, we can use pressure sensors.